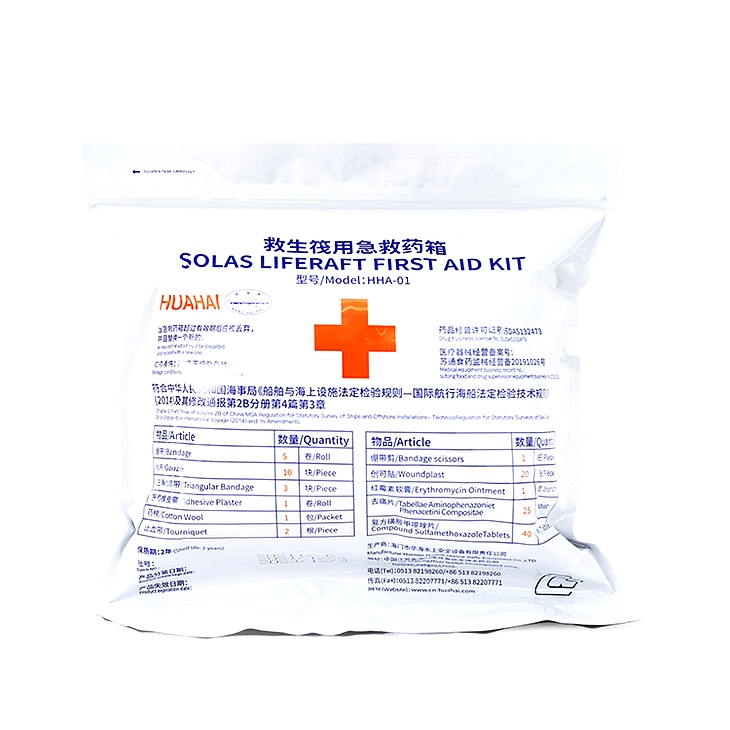
Product number:HHA-01
product name:first aid kit for life raft
Shelf life:2-3 years
Drug Name:3-corner bandage, cotton wool, medical adhesive plaster
Roxithromycin ointment, emergency rescue instructions, etc.
Storage method:dry place
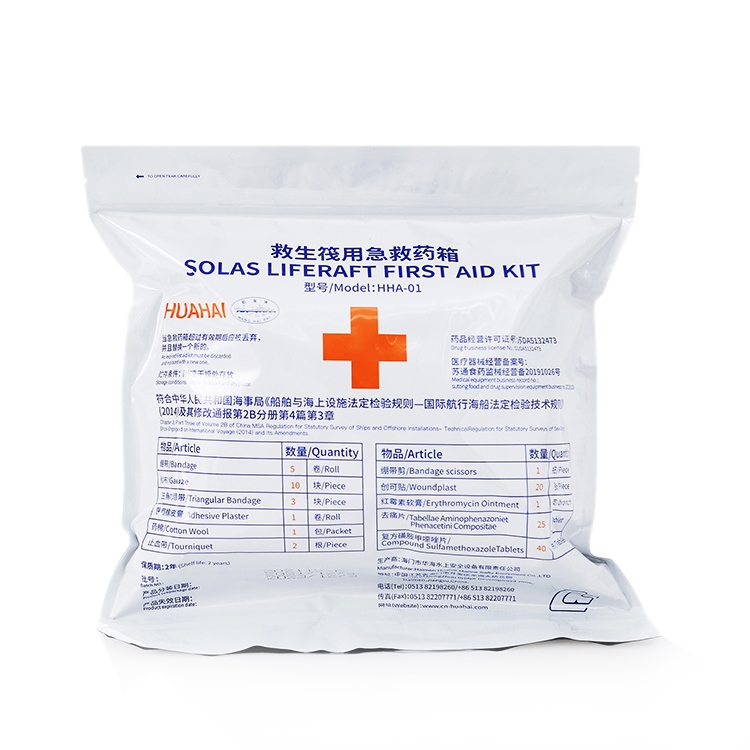
Marine life raft first aid kit medicine details:
Drug Name | Specification | unit | Purpose and usage |
bandage | 4.8x600cm | roll | Bandage to stop bleeding |
gauze | 34x40cm | Piece | bandaging or dressing |
3 corner bandage | Bottom edge 130cm waist 90cm | Piece | Bandaging the injured part of the body |
medical adhesive plaster | 1.2x100cm | roll | for fixing |
cotton wool | 10g | Bag | For disinfection or dressing |
bandage scissors | 10cm | Bundle | Cut bandages or gauze etc. |
tourniquet | 55cm | root | Used for arterial and venous hemostasis, 2-3 circles around the limb, and the blood vessels are compressed by the contraction force of the tourniquet |
Erythromycin ointment | 20g | branch | For water, fire scald antiseptic pain relief, apply to burns |
pain reliever | 0.36g | piece | For antipyretic, analgesic, toothache, cold and fever, etc. Oral: 3 times a day, 1-2 tablets each time |
Compound Sulfamethoxazole Tablets | 0.5g | piece | For respiratory tract, urinary tract, intestinal infection and sepsis, etc. Oral: 2 times a day, 2 tablets each time |
band Aid | 2.5×2cm | open | For emergency treatment of small wounds, directly cover the wound with hemostatic gauze, and paste the plaster on the skin. If the sticky area is contaminated, replace it immediately until the wound heals. |
Special Instructions:
1. There are emergency rescue instructions in each medicine bag, and each product has instructions and precautions for product use.
2. When the first aid kit has expired, it should be discarded and replaced with a new one.
Executive standard:
Comply with Chapter 3, Chapter 4, Chapter 4, Statutory Inspection Rules for Ships and Offshore Installations-Technical Rules for Statutory Inspection of Domestic Seagoing Vessels (2020) of Maritime Safety Administration of the People's Republic of China.
Compliant with the Statutory Inspection Rules for Ships and Offshore Installations of the Maritime Safety Administration of the People's Republic of China - Technical Regulations for Statutory Inspection of Sea-going Ships on International Voyages (2014) and its amendments, Chapter 3, Chapter 4, Volume 2B.
Drug business license number: Su DA5132473
Medical device operation record number: Sutong Food and Drug Administration Equipment Operation No. 20191026